Benedict’s Solution
Last reviewed by Dr. Raj MD on January 12th, 2022.
What is Benedict’s solution?
It is a substance used to test for simple sugars like glucose. It is clear blue solution consists of sodium carbonate, sodium citrate, and copper sulphate. Benedict’s solution is useful in determining whether the sugar is reducible or non-reducible. (1)
What does benedict’s solution test for?
It is one of the commonly used laboratory solution for the detection of various types of sugar as well as checking sugar content in patients with diabetes. It is a quantitative and qualitative test. Before performing benedict’s solution test, it is important to check for the quality of the solution. You have to check the purity of the solution.
To do so, take about 5 ml of benedict’s solution and put inside the test tube. Put the test tube under heat and if it does not change in color, it means that the benedict solution is pure. (1, 2, 3)
Benedict’s Test Procedure
You will need a 5ml of Benedict’s solution placed in a test tube. Add about 5 to 8 drops of the original solution in a test tube filled with benedict’s solution and heat it. Upon boiling, the solution changes in color. If the color remains the same, it means that the sugar content is non-reducible.
A reducible sugar will change the color of the solution into green, yellow, or brick-red. The color depends on the amount of sugar. (3)

Picture 1: An image of a benedict’s solution
Photo Source: www.sciencecompany.com

Photo 2: A result interpretation of Benedict’s test
Picture Source: www.microbiologyinfo.com
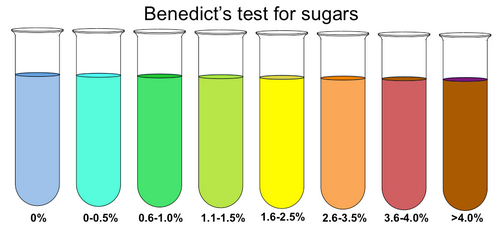 Image 3: Test tubes containing benedict’s solution testing for sugars
Image 3: Test tubes containing benedict’s solution testing for sugars
Picture Source: s3.amazonaws.com
Benedict’s Test Results
Benedict’s solution is used in testing simple carbohydrates. The test helps in identifying reducing sugar such as monosaccharides (glucose, fructose, and galactose) and a few disaccharides (lactose and maltose). They have free ketone. With the help of benedict’s solution, health care professionals will be able to detect the presence of glucose in urine.
What is the implication of reducing sugars? This type of sugar has the ability to transfer hydrogens to other compounds through the process called reduction. The color of the benedict’s solution changes if the reducing sugar is mixed with the Benedict’s reagent and put under a fire. Through Benedict’s test, the sugar in the urine will be detected, which helps in diagnosing diabetes mellitus. (2, 3)
Benedict’s reagent formula is made up of potassium thiocyanate, which helps determine the amount of reducing sugar. It will then form a copper thiocyanate precipitate, which is white in color and useful in titration. For calibration, the titration is repeated using a 1% glucose solution.
As with the benedict’s solution color change, there is an explanation to that. If the benedict’s solution and simple carbohydrates are heated, the benedict’s solution will change its color to orange red or brick red. It is an indication that the property of simple carbohydrate is reduced. The copper ion in the benedict’s solution is reduced, which causes the color to change.
It forms a red copper (I) oxide, which is water insoluble. It will then precipitate out of solution leading to the formation of precipitate. It increases the concentration of reducing sugar. The final color is brick red, which indicates that a greater precipitate is formed. (5, 7)
Some complex carbohydrates like starches don’t come positive with the Benedict’s test. The only way to detect them is breaking them down through digestion and heating. The Benedict’s test for non-reducing sugars such as table sugar is quite different. It needs to decompose the components of sugar into glucose and fructose. That way, the test will come out positive.
Result Interpretation for Benedict’s test
- Green – if the color of the benedict’s solution changes into green, it means that the solution has a sugar percentage of 0.1 to 0.5.
- Yellow – if the color of the solution changes into yellow, there is 0.5 to 1 percent sugar in the solution.
- Orange – An orange color indicates that there is 1 to 1.5% sugar in the solution.
- Red – A sugar percentage of 1.5 to 2 will turn the solution into red.
- Brick red – More than 2% of sugar in the solution will change the color into brick red.
A positive benedict’s test forms a reddish precipitate within three minutes. Glucose is a perfect example of a reducing sugar. If the color of the benedict’s solution remains the same, it means that the solution does not contain reducing sugars. A perfect example of non-reducing sugar is sucrose. (4, 7, 8, 9)
What could possibly affect the result of Benedict’s test?
- Presence of drugs – If certain drugs are present, it could lead to a false positive reaction.
- Presence of chemicals – presence of chemicals in concentrated urine can significantly affect the result of Benedict’s test. Examples are ascorbic acid, creatinine, and urate. (6)
What is a biuret solution?
A biuret solution is usually mistaken to benedict’s solution primarily because they have the same color (clear blue). Benedict’s solution tests sugar while biuret solution tests for the presence of protein in a sample. Changes in the color of the biuret solution will determine the presence of protein.
The sample solution will turn violet if protein is present. A biuret test does not make use of the chemical biuret.
To create a biuret solution, mix in a test tube an equal part of potassium hydroxide and sample material. If the material to be tested is solid, it should be dissolved first. Add a few drops of 1% aqueous solution of copper sulphate. If the color turns violet, it means that the solution is positive of protein.
Some proteins are present in the urine of human. However, if the level of protein in the human urine gets out of control, then it could indicate that there is something wrong with your kidneys. You could also be suffering from high blood pressure, pregnancy, diabetes, heart disease, leukemia, and lupus. (7, 8, 9, 10)
References:
- www.sciencecompany.com
- https://en.wikipedia.org
- www.microbiologyinfo.com
- laboratoryinfo.com
- www.sciencecompany.com
- https://allmedicalstuff.com
- www.quora.com
- www.sigmaaldrich.com
- www.worldofchemicals.com
- Field Confirmation Testing for Suspicious Substances By Rick Houghton